|
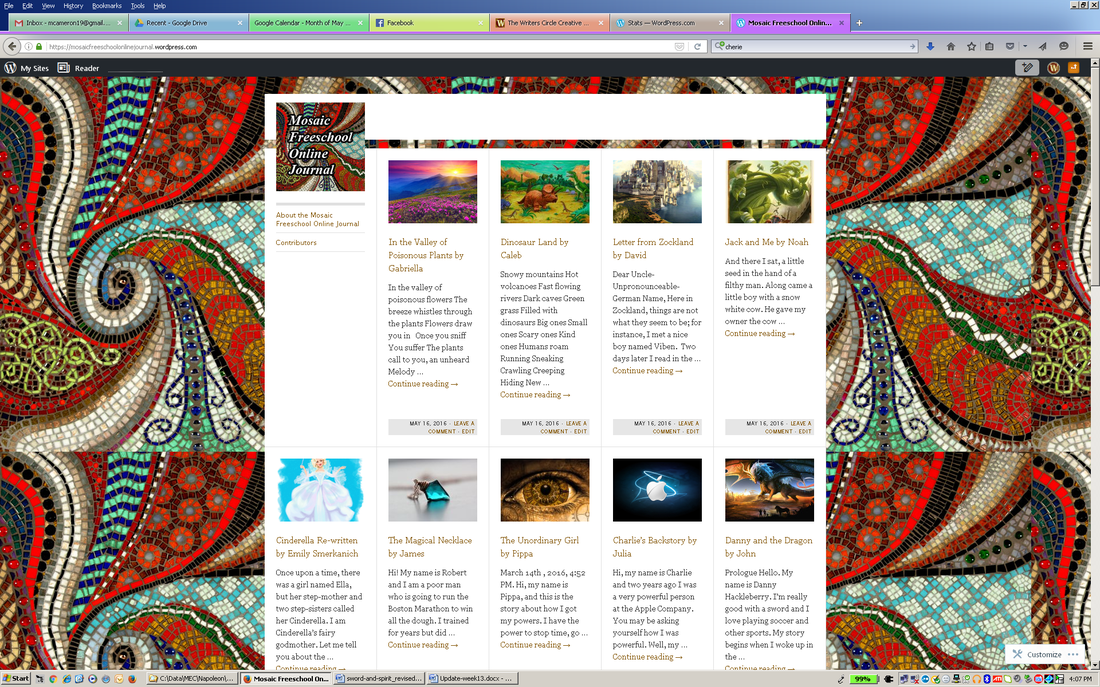
with Michelle Cameron Final Rehearsal and Online JournalToday was our grand rehearsal in the dining room. Each reading was practiced once; each play twice. Do encourage your kids to continue to practice at home. In case they have lost their printed version (which of course some of them will do) – see below! The writers got to use the versions of their writing from our online journal – which is now online! You can visit it here: https://mosaicfreeschoolonlinejournal.wordpress.com. Here’s where you can download and print out their reading if they have lost it. And do feel free to share it with friends and relatives. The kids did a great job! InvitationPlease plan on coming to our reading and play performance next week. We’ll need about 15 minutes to set up, so plan on arriving at 12:30. We’ll be delighted to give voice to our writing from this session – and show you the great job the kids did particularly on their one-act plays!
0 Comments
with Michelle Cameron Play Practice  Today was week two of play practice. Thanks so much to Patti for helping out in class so we could once again rehearse in three separate groups. Some of the kids have already memorized their lines – others still need to do so. They know who they are… Even if they’ve memorized, do encourage them to practice. Next week is our “dress rehearsal” of both our reading pieces and our performances. This will probably take the entire class so that we can work the kinks out. PLEASE do send me a copy of their piece for the reading and our digital publication via email. Here is what I have so far:
If your child’s name is not on this list and you already sent it, please do resend to mcameron@writerscircleworkshops.com THIS WEEK, PLEASE – latest by Saturday. Please send them in either Word on in text – that will help me bring them into our online publication. Magical Objects As the groups were rehearsing, the rest of the students were writing stories that had magical objects in them. Most opted to write about a magical key – it will be interesting next week to hear where they took their stories. It’s amazing how quickly this session is going. We’re all looking forward to our reading/performance! Small Promotional Item Finally, I just wanted to let you all know that some of my own work will be performed in Summit, NJ, on Sunday, May 22, when actors will stage a reading of my verse novel, In the Shadow of the Globe, as a tribute for the 400th anniversary of Shakespeare’s death. Probably not right for the kids in this class, but if you have teens or are yourself interested, I’d love to see you there! You can read more about the performance and register for preferential seating here.
with Michelle Cameron  Today was week one of play practice. Thanks so much to Jayne and Susan for helping out in class so I could have each group rehearse privately. We’ll do this again for the next couple of weeks. The students now have their final scripts. I’ve told them (multiple times) that they are now responsible for hanging on to them. (Of course I’ll bring a couple of extras, anticipating that they won’t.) They’re determined to learn their lines by heart, so they should focus on this during the week. For those students who missed class today, I’ll email the final script home to you. Do look out for the email.  As the groups were rehearsing, the rest of the students were using The Writers Circle’s Story Magic Cards to create new stories. We discussed the fact that some writers write more quickly than others, so those who did not manage to complete their stories in class should take the time this week to do so at home. Don’t worry if these stories don’t make a lot of sense – the cards prompted the kids to add some whacky situations to their stories. Finally, I would like to begin practicing the reading portion of our last class/reading/performance, as well as setting up the digital final publication. Thus far, I only have electronic versions of writing from Gabriella, James, and David. Please send them to me either as a Word document (preferred) or in text. Emily, for instance, submitted hers in Pages format, which I can’t convert. PLEASE HAVE THEM FINALIZE ANY CORRECTIONS AND EMAIL ME THEIR WRITING. Some of them still have some serious rewriting to do – they should know who they are! Email to mcameron@writerscircleworkshops.com, please. I’ll be setting up their online publication over the next few weeks. The kids are excited about the performance – as am I! with Michelle Cameron  Today the groups finished their plays! They read them aloud, and a couple of the groups were able to run through them with props in mind. We’ve left the major props at Mosaic, but each group made a list of any additional props they need. Here it is: Group One:
Conferencing and Revision  I finished my review of everyone’s work EXCEPT for Gabriel and Kaitlyn, both of whom still need to bring the piece they want to read/have published in. Note that it needs to be typed! PLEASE HAVE THEM FINALIZE ANY CORRECTIONS AND EMAIL ME THEIR WRITING. Some of them still have some serious rewriting to do – they should know who they are! Email to mcameron@writerscircleworkshops.com, please. I’ll be setting up their online publication over the next few weeks. Back to Writing Next Week... We’ll be rehearsing a little bit each week until the performance, but I’m eager to get the kids back to writing on their own. If any of them are inspired to write over the week, I’d love it! I gave them a prompt in class: “POW! He (or she) fell down.” Not required, but do encourage them!
with Michelle Cameron A short recap this time... Today we hunkered down to work on our plays. Each of the groups made serious progress, while at least one had some issues with collaboration. It’s hard to write in a group, sometimes! Props and SceneryI’m hoping we’ll be able to finish up our plays next week and start some rehearsals. To that end, I’ve asked the kids to bring in their props and scenery – which in many cases seems to be the most exciting part to them. (Well, they like the writing, too!) Can’t wait to see what they bring! Conferencing and Revision  As they write in their groups, I started bringing them over to show them what I’d like them to do on their final pieces. I didn’t get to everyone, but I will by the end of next week. If I did, please have them revise based on the comments I gave them and bring back both my marked-up copy and their new revision. Please do remember that our last class will be our reading/play performance! We hope to see you all there for that. with Michelle Cameron The play's the thing... For the first time today, we didn’t critique the writing the kids did at home, as I wanted them to launch straight into their plays. We discussed that the back stories they wrote for their characters were chiefly for them to know more about them – and they agreed that it helped them understand them more clearly. A little more pre-planning...Before they launched into the scripts, I asked them to think first about their play’s beginning, middle and end (aka, the plot), and then to dive deeper by defining their scenes. Here’s what they came up with:  Group 1: Beginning: Tiger Lily finishes building CM7A and Charlie tries to buy it. Middle: Charlie plots to steal the robot. End: Charlie hires a man to steal the robot, but he is caught and they find Charlie. Scene 1: Tiger Lily finishes the robot. Scene 2: Charlie reads the newspaper and sees an article about the robot in it. Scene 3: Charlie asks if she can buy the robot but Tiger Lily says no. Scene 4: Charlie plots to capture CM7A. Scene 5: Charlie sneaks through an open window to go into Tiger Lily’s lab/garage. When she gets in, she accidentally knocks down a can. The security guard hears it and arrests her. Scene 6: Charlie is in jail crying with Tiger Lily rejoices  Group 2: Beginning (Scene 1): Zack and Lauren Underwood walk down a street in NY. They decide to make a healing robot for a school science fair. Middle (Scene 2): Zack and Lauren make the robot. They have to test it many times. The robot is perfected on test 71. So they named him Zogar 71. They use a special chip that they invented themselves. End (Scene 3): They are at the science fair. Their teacher, Mrs. Ranch, is not impressed. She eats an apple. Then she chokes on the apple. Zogar 71 gets the apple piece unstuck. Mrs. Ranch is very grateful. Zack, Lauren, and Zagar 71 win the science fair.  Group 3: Beginning: An alien space port Middle: Woods End: a clearing Scene 1: Baby Maddie is lost. Scene 2: Zocz and Roxy battle. Scene 3: Roxy takes Maddie in Scene 4: Old man helps Zocz Scene 5: Roxy sends Maddie away to spy school Scene 6: Maddie befriends Steve at school More to come… They then began writing the actual scripts. These I won’t share, as I hope all of you will attend our reading/play performance during the last class!
They will continue to work on props and scenery at home this week. Again, we don’t need them to come to Mosaic yet! We probably need at least 2 more classes for them to complete writing their plays. While they’re working on this, I want to begin conferencing with them on the piece they’ll read/publish at the end of the session. Please have them type up and bring in the piece they think is best for next class – I’ll start reviewing and working with them as they write! with Michelle Cameron Various and sundry...Today we shared a variety of stories – some still on the power of time, some our fairy tale diaries, and still others just finishing up work we’d started in an earlier class. Among the topics we discussed: the use of very distinct voices when more than one character is telling a story, making sure our reactions are credible (who is that hobo at my door and why would I talk to him?), sticking to either first or third person in a story (not both!), the use of call-backs in a series of stories, the need to create a serious problem (conflict) to resolve, thinking our ideas all the way through, and letting the reader know what they need to know at the appropriate time. Pre-planning - a play! The kids have been talking about writing a play since the first class, and today was week 1 in our playwriting exercise. We divided into three groups: Group 1: Julia, Pippa, James and Noah Group 2: John, Gabriel, and Emily Group 3: Gabrielle, Caleb, David and Kaitlyn  I let them know that one of the characters would be a robot. They decided who that would be, then worked on who the other characters who peopled their play would be. We discussed what you need to know to create a believable character, and they considered their character’s names, what they looked like, what their personality is like, and what they want (though this can have changed based on the plotline.) They then introduced their characters. Next we considered setting and what they would need to stage their play. They became enthusiastic about pulling together props and stage design elements. I warned them that we would NOT be working on this in class but that they certainly can at home. NOTE: we’re not ready for these next week. Please tell them to gather them together and leave them at home. Finally, we got to the part they wanted to dive into from the very beginning: the plot. We have some very different plotlines for our three one-act plays. One of the students in each group acted as the scribe. Here’s what they wrote. (Do reassure them that I’ll bring this back with me next week! Also, excuse any errors of transmission…) Group 1: C-M-7a: this epic glitchy robot is smart and intelligent Tiger Lily – Intelligent workaholic Charlie Gbinge – obsessive buyer – her father is the owner of Dell so Charl… Security officer Costumes: Tiger Lilly – black leggings, white lab coat Charlie – black jeans Props – cardboard Plot: billionaire entrepreneur creates the robot, which Charlie wants to buy. When no one allows that, he decides to steal it instead. Group 2: Robot – Zagar 71 – a healer – power-healer, smart, appearance: silver with buttons, 1 week old Sister – Lauren Underwood – an artist – power-artist, nice, appearance: hair in ponytail, wearing jeans, 15 years old Brother – Zack underwood – a fencer – power-fencing, smart, appearance: flat hair, 14 years old Plot: Lauren and Zack create the robot for a science fair. Their teacher doesn’t like it until Zagar 71 heals her. Group 3: David – alien/robot, 100 years old, Zacz Kaitlyn – a 13-year old girl named Maddie who is fun and kind Gabi – a 16-year old huntress named Roxy who destroyed (half destroyed) Zacz Caleb – BOTH a 15-year old boy named Steve who plays Minecraft and an old man Setting: the kingdom borders a wood. There is sun/stars in an alien planet. Props: laser gun – David bow/arrows – Caleb Lipstick taser – Kaitlyn xbox – Caleb iPad – Caleb big box – anyone Fake deer – Kaitlyn Trees – anyone Plot: Roxy half destroyed Zacz, who is now obsessed with getting revenge. Maddie is a spy. Homework For homework this week, I asked everyone to write their character’s backstory. Next week – we start on the actual script!
with Michelle Cameron Tales of Time We shared our stories about the power of time today. There were some compelling stories – some funny, some extremely gritty and dark. A great many of them were not finished – which is something I want to address below. (Don’t worry - it’s not a problem…) The Use of Emotion in StoriesWe then talked about the fact that different characters in a story will feel differently about a certain event. We used the example of being imprisoned in a dungeon – how the prisoner might feel vs. the emotions of the person who imprisoned them. I also asked if anyone kept a diary. Some do. We discussed the fact that you’ll share your thoughts and emotions in a diary more readily than anywhere else. Then I said – we’re going to write in sprints today – short, concentrated writing in 5-10 minute segments. And I revealed our plot: Oh no! The King has been turned into a dragon! I then revealed the first set of characters that the children wrote diary entries for – the Queen and the Prince. They got to pick which of the two they wanted to write about: They wrote for 10 minutes and shared a couple of the stories. Then we introduced the next two characters – the Evil Magician and the Potion Maker: And then the final pair – the brave knight and the court jester: They didn’t finish the diary entries before I made them move on, but they should have a good enough start to keep going. I’d like all three finished by the time we meet again. Finish and Revise and Piece of Writing In addition, I’d like them to finish a piece of writing that they feel is their best work so far. Please have them type them up and bring them in after the break. This is the start of our revision process before we decide what they are going to publish and/or read at our last meeting. I’m going to start working with them on these pieces in the coming few weeks. Do let them know that they may still opt for something they write later in the class as well!
with Michelle Cameron Fairy Tales Retold  We had some wonderful retelling of fairy tales today:
 There were some particularly fine touches to these stories, including:
The Power of Time I began a discussion of the power of time by asking who had trouble getting up today. I’m sure you won’t be surprised to learn that almost everyone did! So I asked – if you woke up one morning and found you had the power of time, what could you do? Their answers:
They spent some time at the end of class beginning a story in which they (or their characters) have the power of time. I’m looking forward to hearing what they bring in next week! with Michelle Cameron Poetry Reading Because I’d like to hold a reading with your young writers at the end of the session, and because poetry is such a natural for reading aloud (especially when standing) I decided that it was a good time for them to begin to practice. We talked about speaking loudly, slowly, and with expression. We did have some very dramatic readings! It was hard for some of them to pick their heads up and some huddled over the pages. We’ll do this a couple of times before the official reading to get them used to it. Of course, we also critiqued their poems together. Some were quite remarkable! Thanks to all the parents who helped with this exercise. As they worked on their next piece, I called most of them over for a brief one-on-one conference. If your child didn’t get a chance to talk to me, it was mainly because their poems were in their writing notebooks. If those students would type up their poems so I can review them , that would be helpful. Everyone else should have some small suggestions to work through on their poems for next week. Fairy Tales Retold  This week we talked about fairy tales and the fact that all of them are told from the point of view of the heroine or hero – none from the villain’s perspective. We discussed the fact that people don’t wake up in the morning with the idea that “I’m going to do something bad today.” Even villains have a reason for what they do. And sometimes telling a story from a side character’s point of view – such as the pumpkin coach in Cinderella – can make the story totally new. They then started writing their fairy tale from another character’s point of view. Because I met with them on their poems, they may have already finished their story in class. If so, they get to focus on the work on their poem. But if they haven’t finished, they should go ahead and do so for this week’s homework. They were already excited about what they’d written at the end of class and even started sharing with their friends. I’m looking forward to seeing what they’ve done! |
Categories
All
Archives
May 2016
|




















 RSS Feed
RSS Feed